Mouse Anti-Human CD19-FITC (SJ25-C1)
Only %1 left
Cat. No.:
9340-02,
9340-02S
FITC Anti-Human CD19 antibody for use in flow cytometry and immunohistochemistry assays.
As low as
$48.00
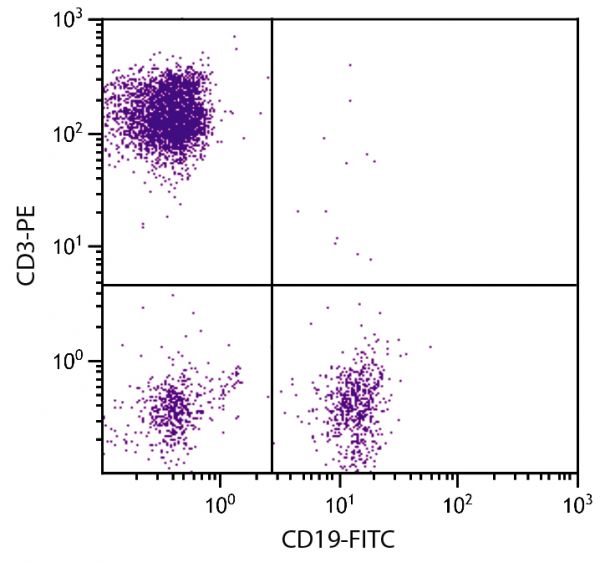

| Clone | SJ25-C1 |
|---|---|
| Isotype | Mouse (BALB/c) IgG1κ |
| Isotype Control | Mouse IgG1-FITC (15H6) |
| Specificity | Human CD19 |
| Alternative Names | B4, B-lymphocyte antigen, Leu-12 |
| Description | CD19 is a type I transmembrane glycoprotein and a member of the immunoglobulin superfamily. It is expressed at all stages of B cell differentiation except terminally differentiated plasma cells. It is also present on the cell surface of follicular dendritic cells. CD19 associates with CD21, CD81, Leu 13 and/or MHC Class II molecules to form a signal transduction complex on the B cell surface. This signaling complex modulates the activation threshold for the B cell antigen receptor (BCR). |
| Immunogen | NALM-1 and NALM-16 leukemia cell line |
| Conjugate | FITC (Fluorescein) |
| Buffer Formulation | Phosphate buffered saline containing < 0.1% sodium azide |
| Clonality | Monoclonal |
| Concentration | Lot specific |
| Volume | 0.25 mL or 1.0 mL |
| Recommended Storage | 2-8°C; Avoid exposure to light |
| Applications |
Flow Cytometry – Quality tested 5-13 Immunohistochemistry-Frozen Sections – Reported in literature 1 Immunoprecipitation – Reported in literature 2 Blocking – Reported in literature 3,4 |
| RRID Number | AB_2796736 |
| Gene ID |
930 (Human) |
| Gene ID Symbol |
CD19 (Human) |
| Gene ID Aliases | B4; CVID3 |
| UniProt ID |
P15391 (Human) |
| UniProt Name |
CD19_HUMAN (Human) |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Vuyyuru R, Liu H, Manser T, Alugupalli KR. Characteristics of Borrelia hermsii infection in human hematopoietic stem cell-engrafted mice mirror those of human relapsing fever. Proc Natl Acad Sci USA. 2011;108:20707-12. (IHC-FS)
- 2. Nadler LM. B cell/leukemia panel workshop: summary and comments. In: Reinherz EL, Haynes BF, Nadler LM, Bernstein ID, editors. Leukocyte Typing II: Human B Lymphocytes. New York: Springer-Verlag; 1986. p. 3-43. (IP)
- 3. Moldenhauer G, Dörken B, Schwartz R, Pezzutto A, Knops J, Hämmerling GJ. Analysis of ten B lymphocyte-specific workshop monoclonal antibodies. In: Reinherz EL, Haynes BF, Nadler LM, Bernstein ID, editors. Leukocyte Typing II: Human B Lymphocytes. New York: Springer-Verlag; 1986. p. 61-7. (Block)
- 4. de Rie MA, Terpstra FG, van Lier RA, von dem Borne AE, Miedema F. Identification of functional epitopes on workshop-defined B-cell membrane molecules. In: McMichael AJ, Beverley PC, Cobbold S, Crumpton MJ, Gilks W, Gotch FM, et al, editors. Leukocyte Typing III: White Cell Differentiation Antigens. Oxford: Oxford University Press; 1987. p. 402-5. (Block)
- 5. Odendahl M, Jacobi A, Hansen A, Feist E, Hiepe F, Burmester GR, et al. Disturbed peripheral B lymphocyte homeostasis in systemic lupus erythematosus. J Immunol. 2000;165:5970-9. (FC)
- 6. Odendahl M, Keitzer R, Wahn U, Hiepe F, Radbruch A, Dörner T, et al. Perturbations of peripheral B lymphocyte homoeostasis in children with systemic lupus erythematosus. Ann Rheum Dis. 2003;62:851-8. (FC)
- 7. Henneken M, Dörner T, Burmester G, Berek C. Differential expression of chemokine receptors on peripheral blood B cells from patients with rheumatoid arthritis and systemic lupus erythematosus. Arthritis Res Ther. 2005;7:R1001-13. (FC)
- 8. Shankar SP, Petrie TA, García AJ, Babensee JE. Dendritic cell responses to self-assembled monolayers of defined chemistries. J Biomed Mater Res A. 2010;92:1487-99. (FC)
- 9. Shankar SP, Babensee JE. Comparative characterization of cultures of primary human macrophages or dendritic cells relevant to biomaterial studies. J Biomed Mater Res A. 2010;92:791-800. (FC)
- 10. Chung BH, Kim KW, Sun IO, Choi SR, Park HS, Jeon EJ, et al. Increased interleukin-17 producing effector memory T cells in the end-stage renal disease patients. Immunol Lett. 2011;141:181-9. (FC)
- 11. Kim KW, Chung BH, Jeon EJ, Kim B, Choi BS, Park CW, et al. B cell-associated immune profiles in patients with end-stage renal disease (ESRD). Exp Mol Med. 2012;44:465-72. (FC)
- 12. Kim HY, Cho M, Jhun JY, Byun JK, Kim E, Yim YB, et al. The imbalance of T helper 17/regulatory T cells and memory B cells during the early post-transplantation period in peripheral blood of living donor liver transplantation recipients. Immunology. 2013;138:124-33. (FC)
- 13. Chung BH, Kim KW, Yu JH, Kim B, Choi BS, Park CW, et al. Decrease of immature B cell and interleukin-10 during early-post-transplant period in renal transplant recipients under tacrolimus based immunosuppression. Transpl Immunol. 2014;30:159-67. (FC)
See All References