Rat Anti-Mouse CD107a-UNLB (1D4B)
Cat. No.:
1920-01
Purified Anti-Mouse CD107a antibody for use in flow cytometry, immunohistochemistry / immunocytochemistry, electron microscopy, western blot, immunoprecipitation, ELISA, and purification assays.
$248.00
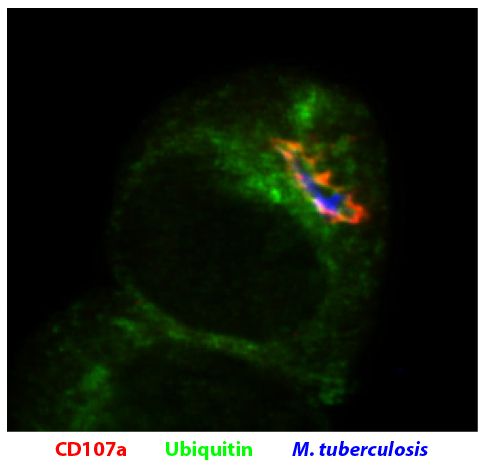
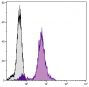
| Clone | 1D4B |
|---|---|
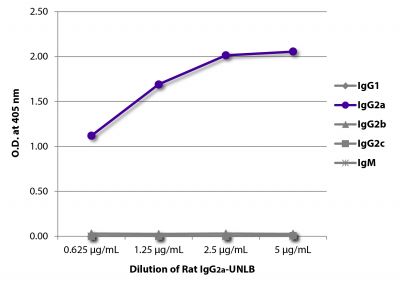
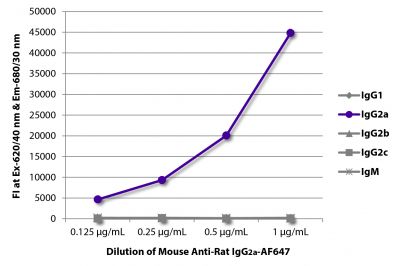
| Isotype | Rat (Sprague Dawley) IgG2aκ |
| Isotype Control | Rat IgG2a-UNLB (KLH/G2a-1-1) |
| Specificity | Mouse CD107a |
| Alternative Names | LAMP-1, lysosomal-associated membrane protein 1 |
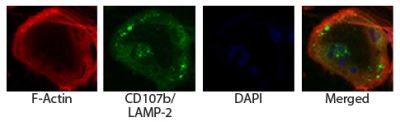
| Description | CD107a, also known as lysosomal-associated membrane protein 1 (LAMP-1), is a heavily glycosylated type I transmembrane protein that constitutes one of the two major sialoglycoproteins on lysosomal membranes that can be used to distinguish lysosomes from other organelles. It is a ligand for galaptin, an S-type lectin present in extracellular matrix, through its recognition of acetyllactosamine oligosaccharide chains and is a ligand for E-selectin-mediated cell adhesion. CD107a is principally expressed in epithelial cells and macrophages in a variety of organs in normal and beige mutant mice. It may function in protecting the inner surface of the lysosomal membrane by forming a barrier to lysosomal hydrolases. |
| Immunogen | Plasma membrane fraction of mouse embryo NIH/3T3 cell line |
| Conjugate | UNLB (Unconjugated) |
| Buffer Formulation | Borate buffered saline, pH 8.2 |
| Clonality | Monoclonal |
| Concentration | 0.5 mg/mL |
| Volume | 1.0 mL |
| Recommended Storage | 2-8°C |
| Applications |
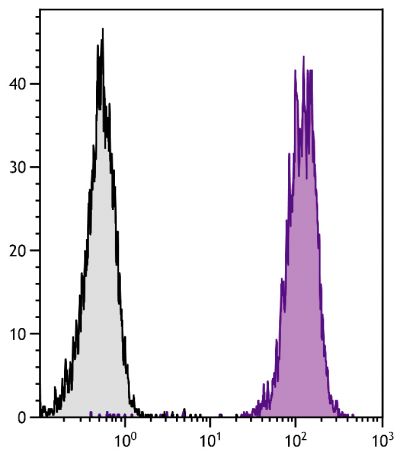
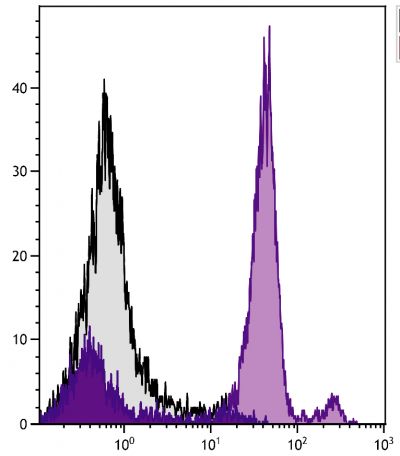
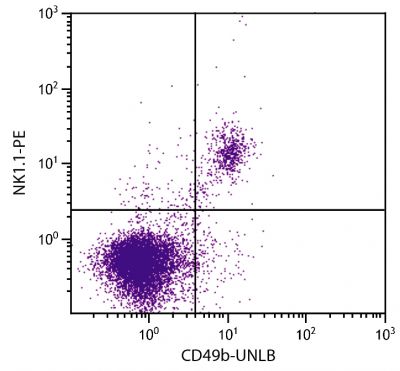
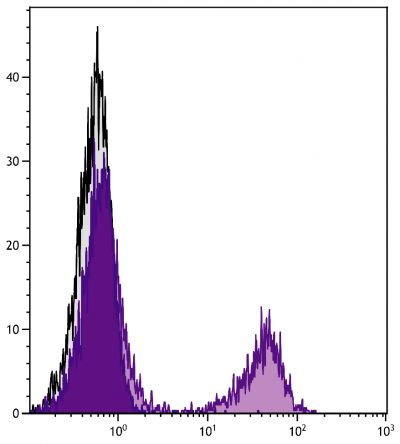
Flow Cytometry – Quality tested 16-18 Immunohistochemistry-Frozen Sections – Reported in literature 3 Immunohistochemistry-Paraffin Sections – Reported in literature 4,5 Immunocytochemistry – Reported in literature 1,6-10 Electron Microscopy – Reported in literature 11 Immunoprecipitation – Reported in literature 1 Western Blot – Reported in literature 12,13 ELISA – Reported in literature 14 Purification – Reported in literature 1,15 |
| RRID Number | AB_2795530 |
| Gene ID |
16783 (Mouse) |
| Gene ID Symbol |
Lamp1 (Mouse) |
| Gene ID Aliases | CD107a; LGP-120; LGP-A; Lamp-1; P2B; Perk |
| UniProt ID |
P11438 (Mouse) |
| UniProt Name |
LAMP1_MOUSE (Mouse) |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Chen JW, Pan W, D'Souza MP, August JT. Lysosome-associated membrane proteins: characterization of LAMP-1 of macrophage P388 and mouse embryo 3T3 cultured cells. Arch Biochem Biophys. 1985;239:574-86. (Immunogen, ICC, IP, Purification)
- 2. Hughes EN, August JT. Characterization of plasma membrane proteins identified by monoclonal antibodies. J Biol Chem. 1981;256:664-71. (Immunogen)
- 3. Milićević NM, Milićević Ž, Miljković MD, Labudović-Borović M, Laan M, Peterson P, et al. Metallophilic macrophages are fully developed in the thymus of autoimmune regulator (Aire)-deficient mice. Histochem Cell Biol. 2009;131:643-9. (IHC-FS)
- 4. Nelson MP, Tse TE, O-Quinn DB, Percival SM, Jaimes EA, Warnock DG, et al. Autophagy-lysosome pathway associated neuropathology and axonal degeneration in the brains of alpha-galactosidase A-deficient mice. Acta Neuropathol Commun. 2014;2:20. (IHC-PS)
- 5. Aula N, Kopra O, Jalanko A, Peltonen L. Sialin expression in the CNS implicates extralysosomal function in neurons. Neurobiol Dis. 2004;15:251-61. (IHC-PS)
- 6. Liu H, Komiya S, Shimizu M, Fukunaga Y, Nagafuchi A. Involvement of p120 carboxy-terminal domain in cadherin trafficking. Cell Struct Funct. 2007;32:127-37. (ICC)
- 7. Hernández-Deviez D, Mackay-Sim A, Wilson JM. A Role for ARF6 and ARNO in the regulation of endosomal dynamics in neurons. Traffic. 2007;8:1750-64. (ICC)
- 8. Song L, Lee C, Schindler C. Deletion of the murine scavenger receptor CD68. J Lipid Res. 2011;52:1542-50. (ICC)
- 9. Hitomi Y, Adachi T, Tsuchiya N, Honda Z, Tokunaga K, Tsubata T. Human CD72 splicing isoform responsible for resistance to systemic lupus erythematosus regulates serum immunoglobulin level and is localized in endoplasmic reticulum. BMC Immunol. 2012;13:72. (ICC)
- 10. Seto S, Tsujimura K, Horii T, Koide Y. Autophagy adaptor protein p62-SQSTM1 and autophagy-related gene Atg5 mediate autophagosome formation in response to Mycobacterium tuberculosis infection in dendritic cells. PLoS One. 2013;8(12):e86017. (ICC)
- 11. Rohrer J, Schweizer A, Russell D, Kornfeld S. The targeting of Lamp1 to lysosomes is dependent on the spacing of its cytoplasmic tail tyrosine sorting motif relative to the membrane. J Cell Biol. 1996;132:565-76. (EM)
- 12. Schneppenheim J, Hüttl S, Mentrup T, Lüllmann-Rauch R, Rothaug M, Engelke M, et al. The intramembrane proteases signal peptide peptidase-like 2a and 2b have distinct functions in vivo. Mol Cell Biol. 2014;34:1398-1411. (WB)
- 13. Bartsch U, Galliciotti G, Jofre GF, Jankowiak W, Hagel C, Braulke T. Apoptotic photoreceptor loss and altered expression of lysosomal proteins in the nclf mouse model of neuronal ceroid lipofuscinosis. Invest Ophthalmol Vis Sci. 2013;54:6952-9. (WB)
- 14. Barnes KA, Mitchell RN. Detection of functional class II-associated antigen: role of a low density endosomal compartment in antigen processing. J Exp Med. 1995;181:1715-27. (ELISA)
- 15. Arterburn LM, Earles BJ, August JT. The disulfide structure of mouse lysosome-associated membrane protein 1. J Biol Chem. 1990;265:7419-23. (Purification)
- 16. Usharauli D, Kamala T. Brief antigenic stimulation generates effector CD8 T cells with low cytotoxic activity and high IL-2 production. J Immunol. 2008;180:4507-13. (FC)
- 17. Halfteck GG, Elboim M, Gur C, Achdout H, Ghadially H, Mandelboim O. Enhanced in vivo growth of lymphoma tumors in the absence of the NK-activating receptor NKp46/NCR1. J Immunol. 2009;182:2221-30. (FC)
- 18. Elboim M, Gazit R, Gur C, Ghadially H, Betser-Cohen G, Mandelboim O. Tumor immunoediting by NKp46. J Immunol. 2010;184:5637-44. (FC)
See All References