Human Type IV Collagen-Solution
Cat. No.:
1250-01S
Purified Human Type IV Collagen solution for use as a coating material and standard.
$371.00
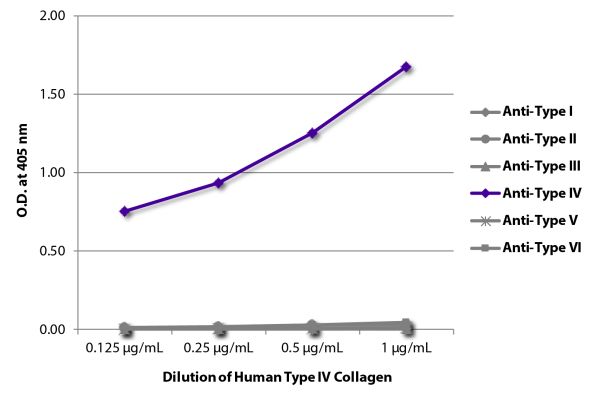

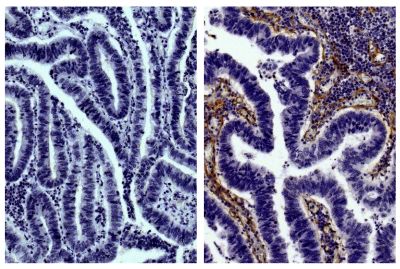
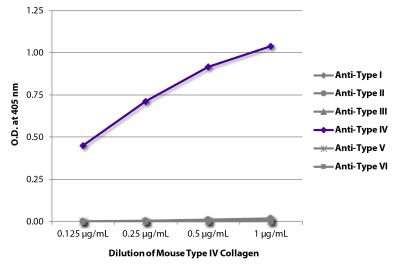
| Description | Collagen is the main structural protein in the extracellular space and is the most abundant protein in the ECM. Collagens are divided into two classes - fibril (types I, II, III, V) and non-fibril (types IV, VI). Type IV collagen is primarily expressed in the basal lamina and therefore constitutes a portion of the basement membrane. Type IV collagen mutations are associated with Alport syndrome and the α3(IV) chain is thought to be the antigen implicated in Goodpasture's syndrome. Type IV collagen is comprised of six isomeric chains designated α1(IV) to α6(IV). |
|---|---|
| Source | Placental villi |
| Purity | > 90% by SDS-PAGE |
| Purification Method | Controlled and limited pepsin digestion followed by selective salt precipitation |
| Buffer Formulation | 500 mM acetic acid |
| Concentration | 0.5 mg/mL |
| Volume | 1.0 mL |
| Recommended Storage | 2-8°C |
| Applications |
ELISA – Quality tested 2-4 SDS-PAGE – Quality tested 1 Coating Material for – Adhesion Studies – Reported in literature 5,6 Blood Disorder Studies – Reported in literature 2-4 Differentiation Studies – Reported in literature 7 ECM Interaction Studies – Reported in literature 8,9 |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Kim T, Moon JH, Savard CE, Kuver R, Lee SP. Effects of lipopolysaccharide on platelet-derived growth factor isoform and receptor expression in cultured rat common bile duct fibroblasts and cholangiocytes. J Gastroenterol Hepatol. 2009;24:1218-25. (SDS-PAGE)
- 2. Doruelo AL, Haberichter SL, Christopherson PA, Boggio LN, Gupta S, Lentz SR, et al. Clinical and laboratory phenotype variability in type 2M von Willebrand disease. J Thromb Haemost. 2017;15:1559-66. (ELISA, Coating, Blood Disorder Studies)
- 3. Garcia J, Flood VH, Haberichter SL, Fahs SA, Mattson JG, Geurts AM, et al. A rat model of severe VWD by elimination of the VWF gene using CRISPR/Cas9. Res Pract Thromb Haemost. 2019;4:64-71. (ELISA, Coating, Blood Disorder Studies)
- 4. Chen W, Voos KM, Josephson CD, Li R. Short-acting anti-VWF (von Willebrand factor) aptamer improves the recovery, survival, and hemostatic functions of refrigerated platelets. Arterioscler Thromb Vasc Biol. 2019;39:2028-37. (ELISA, Coating, Blood Disorder Studies)
- 5. Midwood KS, Salter DM. NG2/HMPG modulation of human articular chondrocyte adhesion to type VI collagen is lost in osteoarthritis. J Pathol. 2001;195:631-5. (Coating, Adhesion Studies)
- 6. Imakawa K, Imai M, Sakai A, Suzuki M, Nagaoka K, Sakai S, et al. Regulation of conceptus adhesion by endometrial CXC chemokines during the implantation period in sheep. Mol Reprod Dev. 2006;73:850-8. (Coating, Adhesion Studies)
- 7. Hohn H, Denker H. The role of cell shape for differentiation of choriocarcinoma cells on extracellular matrix. Exp Cell Res. 1994;215:40-50. (Coating, Differentiation Studies)
- 8. Bidanset DJ, Guidry C, Rosenberg LC, Choi HU, Timpl R, Hook M. Binding of the proteoglycan decorin to collagen type VI. J Biol Chem. 1992;267:5250-6. (Coating, ECM Interaction Studies)
- 9. Liu Z, Han X, Chen R, Zhang K, Li Y, Fruge S, et al. Microfluidic mapping of cancer cell-protein binding interaction. ACS Appl Mater Interfaces. 2017;9:22143-8. (Coating, ECM Interaction Studies)
See More