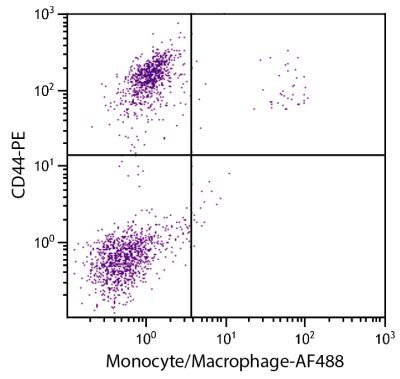
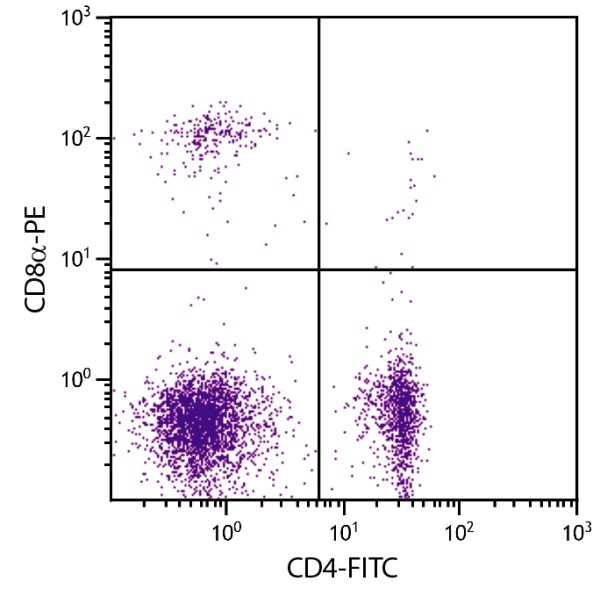
Mouse Anti-Chicken CD8α-PE (EP72)
Cat. No.:
8390-09
PE Anti-Chicken CD8α antibody for use in flow cytometry assays.
$262.00

| Clone | EP72 |
|---|---|
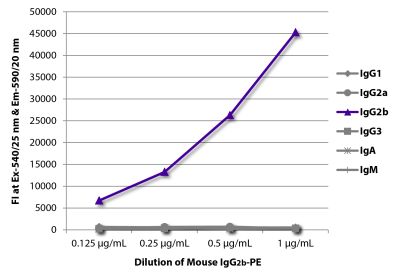
| Isotype | Mouse (BALB/c) IgG2bκ |
| Isotype Control | Mouse IgG2b-PE (A-1) |
| Specificity | Chicken CD8α |
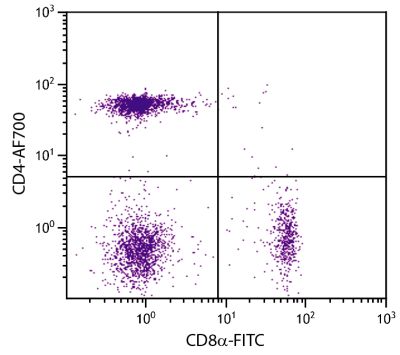
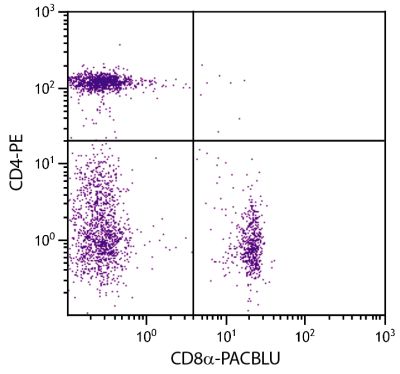
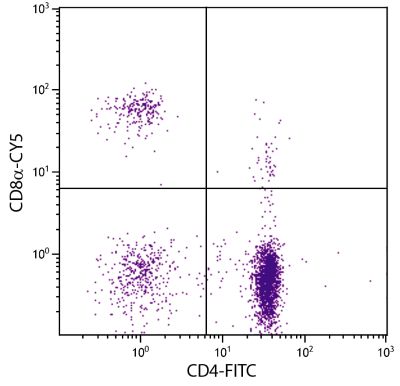
| Description | In the chicken, the CD8 molecule is present in two forms - (i) a homodimer of two α chains and (ii) a heterodimer of an α chain and a β chain. Chicken CD8 is expressed on approximately 80% of thymocytes, 15% of blood mononuclear cells and 50% of spleen cells but less than 1% of cells in the bursa and bone marrow. While the vast majority of CD8+ cells in the thymus, spleen, and blood of adult chickens express both CD8α- and CD8β-chains, a relatively large proportion of the CD8+ TCRγδ cells in the spleens of embryos and young chicks express only the α-chain of CD8. Among intestinal epithelial lymphocytes, the major CD8+ T cell populations present in mice are conserved but there is a population of TCRγδ CD8αβ cells in the chicken that is not found in rodents. The monoclonal antibody EP72 recognizes the CD8α chain. |
| Immunogen | Chicken splenocytes |
| Conjugate | PE (R-phycoerythrin) |
| Buffer Formulation | Phosphate buffered saline containing < 0.1% sodium azide and a stabilizer |
| Clonality | Monoclonal |
| Concentration | 0.1 mg/mL |
| Volume | 1.0 mL |
| Recommended Storage | 2-8°C; Avoid exposure to light; Do not freeze |
| Applications |
Flow Cytometry – Quality tested 4-7 Immunohistochemistry-Frozen Sections – Reported in literature 2,3 |
| RRID Number | AB_2796540 |
| Gene ID |
403158 (Chicken) |
| Gene ID Symbol |
CD8A (Chicken) |
| Gene ID Aliases | CD8 |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Paramithiotis E, Tkalec L, Ratcliffe MJ. High levels of CD45 are coordinately expressed with CD4 and CD8 on avian thymocytes. J Immunol. 1991;147:3710-7. (Immunogen)
- 2. Reemers SS, van Haarlem D, Groot Koerkamp MJ, Vervelde L. Differential gene-expression and host-response profiles against avian influenza virus within the chicken lung due to anatomy and airflow. J Gen Virol. 2009;90:2134-46. (IHC-FS)
- 3. Reemers SS, Jansen C, Groot Koerkamp MJ, van Haarlem D, van de Haar P, Degen WG, et al. Reduced immune reaction prevents immunopathology after challenge with avian influenza virus: a transcriptomics analysis of adjuvanted vaccines. Vaccine. 2010;28:6351-60. (IHC-FS)
- 4. Marmor MD, Benatar T, Ratcliffe MJ. Retroviral transformation in vitro of chicken T cells expressing either α/β or γ/δ T cell receptors by reticuloendotheliosis virus strain T. J Exp Med. 1993;177:647-56. (FC)
- 5. Choi KD, Lillehoj HS, Song KD, Han JY. Molecular and functional characterization of chicken IL-15. Dev Comp Immunol. 1999;23:165-77. (FC)
- 6. Chen KL, Tsay SM, Chiou PW, Sun CP, Weng BC. Effects of caponization and different forms of exogenous androgen implantation on immunity in male chicks. Poult Sci. 2010;89:887-94. (FC)
- 7. Norup LR, Dalgaard TS, Pedersen AR, Juul-Madsen HR. Assessment of Newcastle disease-specific T cell proliferation in different inbred MHC chicken lines. Scand J Immunol. 2011;74:23-30. (FC)
See More