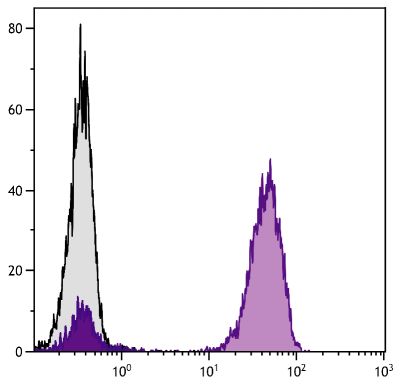
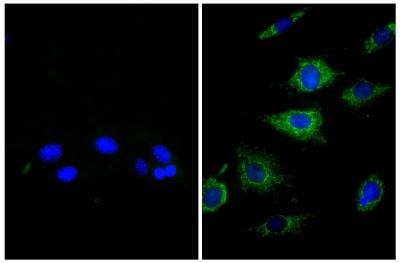
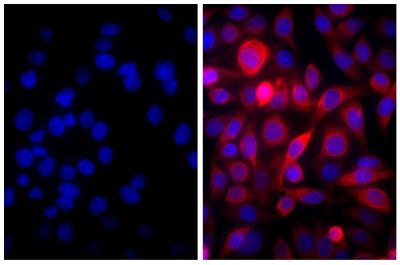
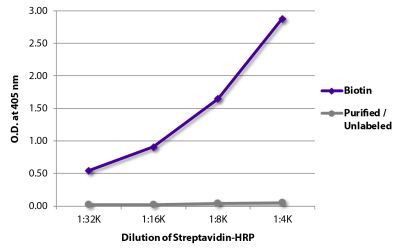
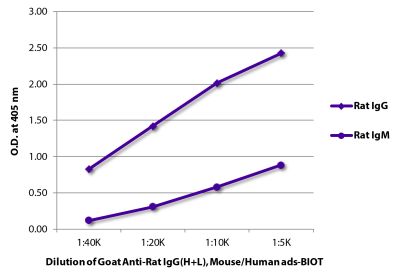
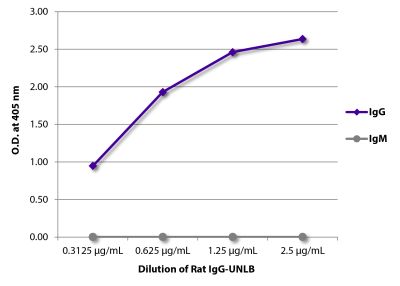
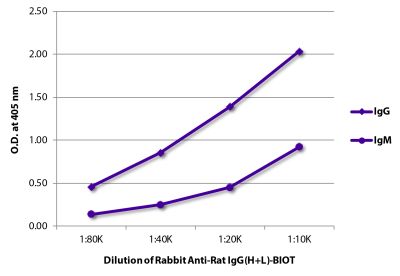
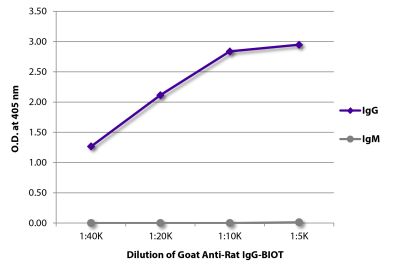
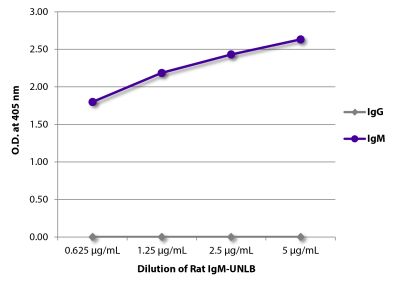
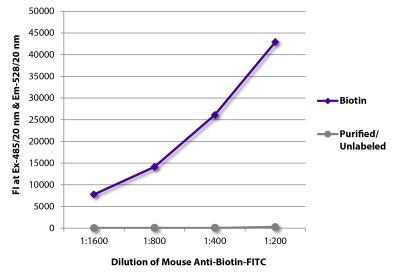
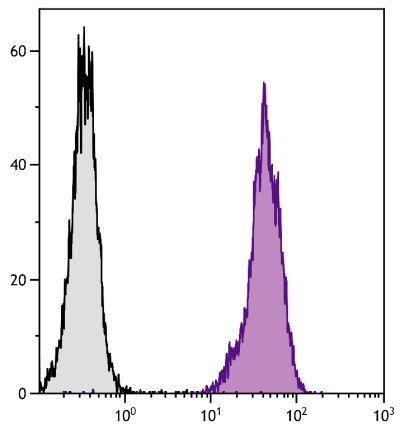
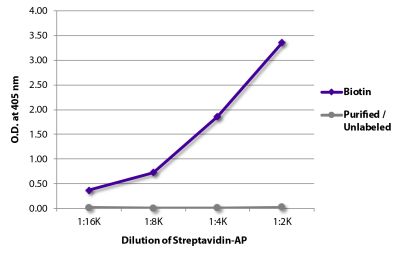
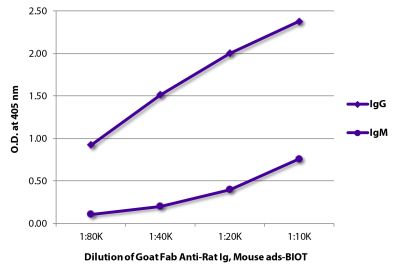
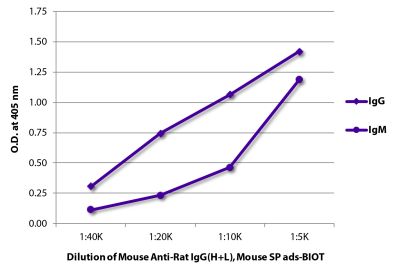
Goat Anti-Rat IgG(H+L), Mouse ads-BIOT
Cat. No.:
3050-08
Goat Anti-Rat IgG(H+L)-Biotin antibody with minimal reactivity to mouse proteins for use in ELISA, flow cytometry, immunohistochemistry / immunocytochemistry, western blot, and separation assays.
$192.00


| Isotype | Goat IgG |
|---|---|
| Isotype Control | Goat IgG-BIOT |
| Specificity | Reacts with the heavy and light chains of rat IgG and the light chains of rat IgM |
| Source | Pooled antisera from goats hyperimmunized with rat IgG |
| Cross Adsorption | Mouse immunoglobulins and pooled sera; may react with immunoglobulins from other species |
| Purification Method | Affinity chromatography on pooled rat IgG covalently linked to agarose |
| Conjugate | BIOT (Biotin) |
| Buffer Formulation | Phosphate buffered saline containing < 0.1% sodium azide |
| Clonality | Polyclonal |
| Concentration | 0.5 mg/mL |
| Volume | 2.0 mL |
| Recommended Storage | 2-8°C |
| Applications |
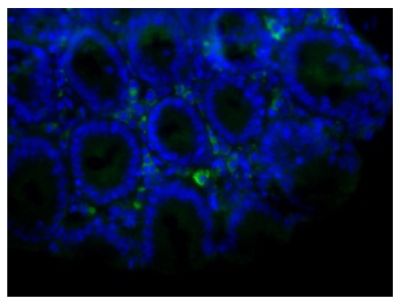
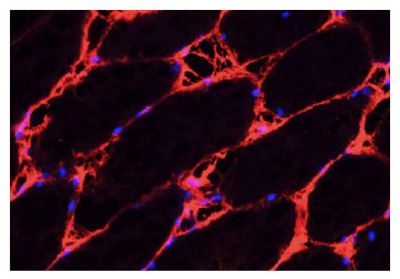
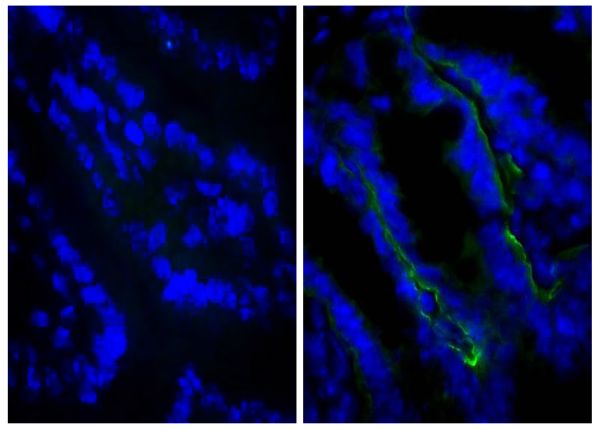
Quality tested applications for relevant formats include - ELISA 1-4 FLISA Flow Cytometry 5-10 Other referenced applications for relevant formats include - Immunohistochemistry-Frozen Sections 11-17 Immunohistochemistry-Paraffin Sections 18-20 Imuunocytochemistry 21,22 Western Blot 23-25 Separation 4 |
| RRID Number | AB_2795833 |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Putterman C, Diamond B. Immunization with a peptide surrogate for double-stranded DNA (dsDNA) induces autoantibody production and renal immunoglobulin deposition. J Exp Med. 1998;188:29-38. (ELISA)
- 2. Yu KO, Im JS, Illarianov PA, Ndonye RM, Howell AR, Besra GS, et al. Production and characterization of monoclonal antibodies against complexes of the NKT cell ligand α-galactosylceramide bound to mouse CD1d. J Immunol Methods. 2007;323:11-23. (ELISA)
- 3. Swanson CL, Wilson TJ, Strauch P, Colonna M, Pelanda R, Torres RM. Type I IFN enhances follicular B cell contribution to the T cell-independent antibody response. J Exp Med. 2010;207:1485-1500. (ELISA)
- 4. Hansen RJ, Brown RM, Lu J, Wroblewski VJ. Qualification of a free ligand assay in the presence of anti-ligand antibody Fab fragments. Mabs. 2013;5:288-96. (ELISA, Sep)
- 5. Pittet MJ, Zippelius A, Speiser DE, Assenmacher M, Guillaume P, Valmori D, et al. Ex vivo IFN-γ secretion by circulating CD8 T lymphocytes: implications of a novel approach for T cell monitoring in infectious and malignant diseases. J Immunol. 2001;166:7634-40. (FC)
- 6. Rizzardi GP, Harari A, Capiluppi B, Tambussi G, Ellefsen K, Ciuffreda D, et al. Treatment of primary HIV-1 infection with cyclosporin A coupled with highly active antiretroviral therapy. J Clin Invest. 2002;109:681-8. (FC)
- 7. Choi K, Kim J, Lee Y, Kim J, Suh B, Kim H, et al. Concurrent delivery of GM-CSF and B7-1 using an oncolytic adenovirus elicits potent antitumor effect. Gene Ther. 2006;13:1010-20. (FC)
- 8. Bossen C, Ingold K, Tardivel A, Bodmer J, Gaide O, Hertig S, et al. Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J Biol Chem. 2006;281:13964-71. (FC)
- 9. Ronconi E, Sagrinati C, Angelotti ML, Lazzeri E, Mazzinghi B, Ballerini L, et al. Regeneration of glomerular podocytes by human renal progenitors. J Am Soc Nephrol. 2009;20:322-32. (FC)
- 10. Long MA, Rossi FM. Targeted cell fusion facilitates stable heterokaryon generation in vitro and in vivo. PloS One. 2011;6:e26381. (FC)
- 11. Csencsits KL, Jutila MA, Pascual DW. Nasal-associated lymphoid tissue: phenotypic and functional evidence for the primary role of peripheral node addressin in naive lymphocyte adhesion to high endothelial venules in a mucosal site. J Immunol. 1999;163:1382-9. (IHC-FS)
- 12. Manley HA, Lennon VA. Endoplasmic reticulum membrane-sorting protein of lymphocytes (BAP31) is highly expressed in neurons and discrete endocrine cells. J Histochem Cytochem. 2001;49:1235-43. (IHC-FS)
- 13. Yu P, Wang Y, Chin RK, Martinez-Pomares L, Gordon S, Kosco-Vibois MH, et al. B cells control the migration of a subset of dendritic cells into B cell follicles via CXC chemokine ligand 13 in a lymphotoxin-dependent fashion. J Immunol. 2002;168:5117-23. (IHC-FS)
- 14. Maehr R, Kraus M, Ploegh HL. Mice deficient in invariant-chain and MHC class II exhibit a normal mature B2 cell compartment. Eur J Immunol. 2004;34:2230-6. (IHC-FS)
- 15. He X, Schoeb TR, Panoskaltsis-Mortari A, Zinn KR, Kesterson RA, Zhang J, et al. Deficiency of P-selectin or P-selectin glycoprotein ligand-1 leads to accelerated development of glomerulonephritis and increased expression of CC chemokine ligand 2 in lupus-prone mice. J Immunol. 2006;177:8748-56. (IHC-FS)
- 16. El Shikh ME, El Sayed RM, Wu Y, Szakal AK, Tew JG. TLR4 on follicular dendritic cells: an activation pathway that promotes accessory activity. J Immunol. 2007;179:4444-50. (IHC-FS)
- 17. Ubogu EE, Yosef N, Xia RH, Sheikh KA. Behavioral, electrophysiological, and histopathological characterization of a severe murine chronic demyelinating polyneuritis model. J Peripher Nerv Syst. 2012;17:53-61. (IHC-FS)
- 18. Cattoretti G, Angelin-Duclos C, Shaknovich R, Zhou H, Wang D, Alobeid B. PRDM1/Blimp-1 is expressed in human B-lymphocytes committed to the plasma cell lineage. J Pathol. 2005;206:76-86. (IHC-PS)
- 19. Lunsford KE, Horne PH, Koester MA, Eiring AM, Walker JP, Dziema HL, et al. Activation and maturation of alloreactive CD4-independent, CD8+ cytolytic T cells. Am J Transplant. 2006;6:2268-81. (IHC-PS)
- 20. van den Borne P, Rygiel TP, Hoogendoorn A, Westerlaken GH, Boon L, Quax PH, et al. The CD200-CD200 receptor inhibitory axis controls arteriogenesis and local T lymphocyte influx. PLoS One. 2014;9(6):e98820. (IHC-PS)
- 21. Elstner E, Linker-Israeli M, Said J, Umiel T, de Vos S, Shintaku IP, et al. 20-epi-vitamin D3 analogues: a novel class of potent inhibitors of proliferation and inducers of differentiation of human breast cancer cell lines. Cancer Res. 1995;55:2822-30. (ICC)
- 22. Chung Y, Zhang N, Wooten RM. Borrelia burgdorferi elicited-IL-10 suppresses the production of inflammatory mediators, phagocytosis, and expression of co-stimulatory receptors by murine macrophages and/or dendritic cells. PLoS One. 2013;8(12):e84980. (ICC)
- 23. Tran TM, Satumtira N, Dorris ML, May E, Wang A, Furuta E, et al. HLA-B27 in transgenic rats forms disulfide-linked heavy chain oligomers and multimers that bind to the chaperone BiP. J Immunol. 2004;172:5110-9. (WB)
- 24. Ohta K, Mizuno A, Ueda M, Li S, Suzuki Y, Hida Y, et al. Autophagy impairment stimulates PS1 expression and γ-secretase activity. Autophagy. 2010;6:345-52. (WB)
- 25. Rice DS, Hansen GM, Liu F, Crist MJ, Newhouse MM, Potter D, et al. Keratinocyte migration in the developing eyelid requires LIMK2. PLoS One. 2012;7(10):e47168. (WB)
See More