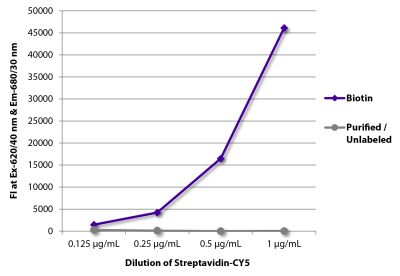
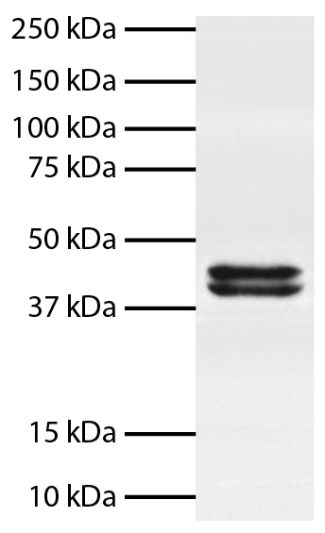
Rat Anti-Mouse TNF-α-BIOT (MP6-XT3)
Cat. No.:
10229-08
Biotin Anti-Mouse TNF-α antibody for use in flow cytometry, immunohistochemistry, western blot, ELISA, ELISpot, and multiplex assays.
$266.00
| Clone | MP6-XT3 |
|---|---|
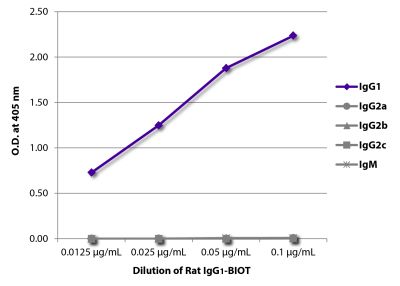
| Isotype | Rat IgG1κ |
| Isotype Control | Rat IgG1-BIOT (KLH/G1-2-2) |
| Specificity | Mouse TNF-α |
| Alternative Names | Tumor necrosis factor-α, macrophage cytotoxic factor , MCF, differentiation inducing factor, DIF, cachectin, necrosin, TNFSF-2 |
| Immunogen | E. coli-expressed mouse TNF-α |
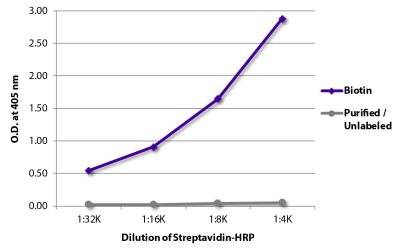
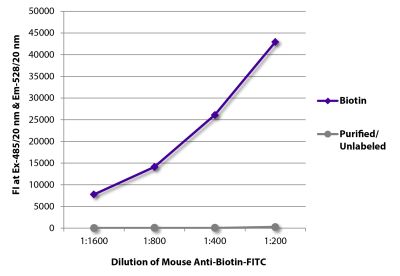
| Conjugate | BIOT (Biotin) |
| Buffer Formulation | Phosphate buffered saline containing < 0.1% sodium azide |
| Clonality | Monoclonal |
| Concentration | 0.5 mg/mL |
| Volume | 1.0 mL |
| Recommended Storage | 2-8°C |
| Applications |
ELISA-Detection – Quality tested 2 ELISA-Capture – Reported in literature 1,3 ELISpot-Capture – Reported in literature 4 ELISpot-Detection – Reported in literature 5 Flow Cytometry – Reported in literature 5-7 Immunohistochemsitry-Frozen Sections – Reported in literature 8-13 Immunohistochemsitry-Paraffin Sections – Reported in literature 14 Western Blot – Reported in literature 15 Neutralization – Reported in literature 1,16,17 Multiplex-Detection – Reported in literature 2 |
| RRID Number | AB_2794271 |
| Gene ID |
21926 (Mouse) |
| Gene ID Symbol |
Tnf (Mouse) |
| Gene ID Aliases | DIF; Tnfa; TNF-a; TNFSF2; Tnlg1f; Tnfsf1a; TNFalpha; TNF-alpha |
| UniProt ID |
P06804 (Mouse) |
| UniProt Name |
TNFA_MOUSE (Mouse) |
Documentation
Certificate of Analysis Lookup
Enter the Catalog Number and Lot Number for the Certificate of Analysis you wish to view
- 1. Abrams JS, Roncarolo M, Yssel H, Andersson U, Gleich GJ, Silver JE. Strategies of anti-cytokine monoclonal antibody development: immunoassay of IL-10 and IL-5 in clinical samples. Immunol Rev. 1992;127:5-24. (ELISA-Capture, Neut)
- 2. Carson RT, Vignali DA. Simultaneous quantitation of 15 cytokines using a multiplexed flow cytometric assay. J Immunol Methods. 1999;227:41-52. (ELISA-Detection, Multiplex-Detection)
- 3. Abrams JS. Immunoenzymetric assay of mouse and human cytokines using NIP-labeled anti-cytokine antibodies. Curr Protoc Immunol. 2001;6.20:1-15. (ELISA-Capture)
- 4. Bordmann G, Rudin W, Favre N. Immunization of mice with phosphatidylcholine drastically reduces the parasitaemia of subsequent Plasmodium chabaudi chabaudi blood-stage infections. Immunology. 1998;94:35-40.(ELISPOT-Capture)
- 5. Korten S, Anderson RJ, Hannan CM, Sheu EG, Sinden R, Gadola S, et al. Invariant Vα14 chain NKT cells promote Plasmodium berghei circumsporozoite protein-specific gamma interferon- and tumor necrosis factor alpha-producing CD8+ T cells in the liver after poxvirus vaccination of mice. Infect Immun. 2005;73:849-58. (ELISPOT-Detection, FC)
- 6. Zajac AJ, Quinn DG, Cohen PL, Frelinger JA. Fas-dependent CD4+ cytotoxic T-cell-mediated pathogenesis during virus infection. Proc Natl Acad Sci USA. 1996;93:14730-5. (FC)
- 7. Feng H, Zhang D, Palliser D, Zhu P, Cai S, Schlesinger A, et al. Listeria-infected myeloid dendritic cells produce IFN-β, priming T cell activation. J Immunol. 2005;175:421-32. (FC)
- 8. Henke A, Huber S, Stelzner A, Whitton JL. The role of CD8+ T lymphocytes in coxsackievirus B3-induced myocarditis. J Virol. 1995;69:6720-8. (IHC-FS)
- 9. Martinelli TM, Van Driel IR, Alderuccio F, Gleeson PA, Toh B. Analysis of mononuclear cell infiltrate and cytokine production in murine autoimmune gastritis. Gastroenterology. 1996;110:1791-802. (IHC-FS)
- 10. Morris MM, Dyson H, Baker D, Harbige LS, Fazakerley JK, Amor S. Characterization of the cellular and cytokine response in the central nervous system following Semliki Forest virus infection. J Neuroimmunol. 1997;74:185-97. (IHC-FS)
- 11. Hersmann GH, Kriegsmann J, Simon J, Hüttich C, Bräuer R. Expression of cell adhesion molecules and cytokines in murine antigen-induced arthritis. Cell Adhes Commun. 1998;6:69-82. (IHC-FS)
- 12. Tsao N, Kanakamma PP, Luh T, Chou C, Lei H. Inhibition of Escherichia coli-induced meningitis by carboxyfullerence. Antimicrob Agents Chemother. 1999;43:2273-7. (IHC-FS)
- 13. Tsao N, Hsu HP, Wu CM, Liu CC, Lei HY. Tumour necrosis factor-α causes an increase in blood-brain barrier permeability during sepsis. J Med Microbiol. 2001;50:812-21. (IHC-FS)
- 14. Huang T, Tsai S, Liu L, Liu YL, Liu H, Chuang KP. Effect of Arctium lappa L. in the dextran sulfate sodium colitis mouse model. World J Gastroenterol. 2010;16:4193-9. (IHC-PS)
- 15. Rogove AD, Lu W, Tsirka SE. Microglial activation and recruitment, but not proliferation, suffice to mediate neurodegeneration. Cell Death Differ. 2002;9:801-6. (WB)
- 16. Grohmann U, Van Snick J, Campanile F, Silla S, Giampietri A, Vacca C, et al. IL-9 protects mice from Gram-negative bacterial shock: suppression of TNF-α, IL-12, and IFN-γ, and induction of IL-10. J Immunol. 2000;164:4197-203. (Neut)
- 17. Ghorpade DS, Holla S, Kaveri SV, Bayry J, Patil SA, Balaji KN. Sonic hedgehog-dependent induction of microRNA 31 and microRNA 150 regulates Mycobacterium bovis BCG-driven toll-like receptor 2 signaling. Mol Cell Biol. 2013;33:543-56. (Neut)
See More